Picture this: A popular celebrity trend turns into a national legal storm—and suddenly, what seemed glamorous is surrounded by serious concerns. That’s the reality with Ozempic. Once lauded as “the magic weight-loss pen,” it has now become the focal point of a sprawling multidistrict litigation (MDL). This isn’t about sensationalism—it’s about understanding how real risks, media influence, research priorities, and public health intersect.
What’s Going On in the Courtroom?
As of August 2025, the legal battle over Ozempic has grown into a massive MDL—MDL No. 3094—centralized in the Eastern District of Pennsylvania, under U.S. District Judge Karen S. Marston. Originally overseen by Judge Gene E. K. Pratter until her untimely passing in May 2024, the MDL now includes over 2,190 pending lawsuits by August, according to law firm tracking Federal Lawyer+12Lawsuit Information Center+12Verus LLC+12.
Plaintiffs allege that Novo Nordisk, and others producing similar semaglutide-based GLP‑1 receptor agonists (like Wegovy, Rybelsus, as well as Eli Lilly’s Mounjaro), failed to inform users about serious potential side effects including gastroparesis, ileus, pancreatitis, and even vision loss (NAION—non‑arteritic anterior ischemic optic neuropathy) New York Injury Law News+11Lawsuit Information Center+11Melinda J. Helbock, A.P.C. San Diego+11.
Why MDL matters: Bringing hundreds—or thousands—of related lawsuits into one venue streamlines discovery, expert testimony, and trial planning. It’s a faster, more efficient judicial approach than dozens of separate trials dragging on across the country paed.uscourts.govMelinda J. Helbock, A.P.C. San Diego.
The Legal Machinery in Motion
- Leadership structure: Judge Pratter formed a robust plaintiffs’ leadership committee, including notable firms such as Motley Rice, Seeger Weiss, Morgan & Morgan, and others paed.uscourts.gov+15Verus LLC+15Reuters+15.
- Key hurdles: Judge Marston has taken proactive steps—ruling early on diagnostic thresholds for gastroparesis and the adequacy of Ozempic’s warning labels, a move that aims to streamline the litigation but has sparked debate about undermining individual cases Reuters+14Reuters+14Reuters+14.
- Special tracks: Plaintiffs have requested a specialized “Vision Injury Track” for NAION claims, distinguishing them from gastrointestinal (GI) claims to address their unique scientific and factual challenges Motley Rice+3Lawsuit Information Center+3Federal Lawyer+3.
- Bellwether trials ahead: As cases pile up, the court is gearing up for representative bellwether trials—crucial test cases that could shape settlement discussions and trial timelines. Legal analysts predict full MDL resolution could take 12–36 months, with settlements possibly looming if early trials indicate strong evidence Goldwater Law Firm+5Melinda J. Helbock, A.P.C. San Diego+5Melinda J. Helbock, A.P.C. San Diego+5.
Beyond the Court: Celebrity Influence vs. Scientific Rigor
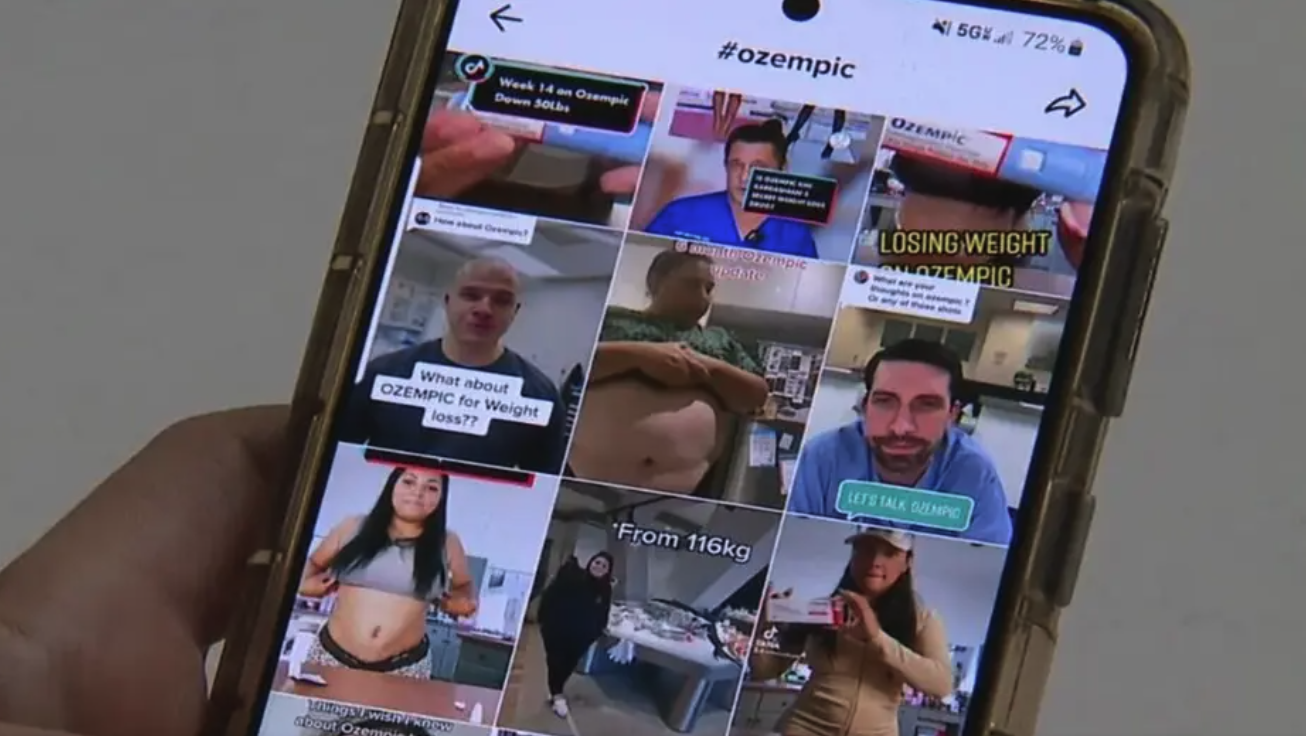
Let’s not lose sight of why this happened in the first place. Ozempic’s meteoric rise coincided with celebrity trends and lifestyle narratives. TikTok posts, high-profile endorsements, and Instagram “before-and-after” reels fueled demand. Almost overnight, semaglutide became the “celebrity magic formula.”
But while media buzz can spark widespread interest, it doesn’t equal medical consensus. Serious side effects—despite being noted in clinical labeling—didn’t become front-page news until lawsuits surfaced. This serves a valuable lesson: enthusiasm should not outpace evidence.
Nexos Pharma’s Perspective: Research-Only Semaglutide vs. Celebrity Hype

At Nexos Pharma, our semaglutide offerings are strictly designated for research purposes only. There’s a fundamental distinction: we’re facilitating scientific inquiry, not promoting weight-loss narratives. Semaglutide—whether branded or generically produced—is the same molecule. The difference is in how it’s presented and used.
We don’t claim Ozempic is ineffective or harmful; rather, we emphasize thoughtful research backed by transparent data. While some may chase celebrity-driven promises, researchers understand that deeper investigation into mechanisms, dosing, and long-term outcomes paints a more reliable picture.
A Deeper View: Risks, Regulatory Signals, and Real-World Evidence
- Gastrointestinal risks: Many lawsuits involve gastroparesis—delayed stomach emptying—argued to be underdisclosed medically paed.uscourts.govReuters.
- Vision concerns: Ophthalmologists and attorneys cite growing evidence tying semaglutide use to sudden vision issues, with over 140 cases now referencing partial or total blindness Lawsuit Information Center+2Peterson & Associates, P. C.+2.
- Expanding claims: Additional injuries like pancreatitis, kidney injury, and even hair loss (suggested in dermatological reports) are under evaluation for inclusion Melinda J. Helbock, A.P.C. San Diego.
- Regulatory watchers: There’s mounting pressure on agencies like the FDA and EMA to update warning labels—especially concerning vision and severe GI risks Lawsuit Information Center.
What This Means for Researchers and Curious Readers
- Celebrity endorsement can inspire, but research demands scrutiny.
- MDL outcomes influence public policy, regulatory guidance, and medical practice—far beyond courtroom awards.
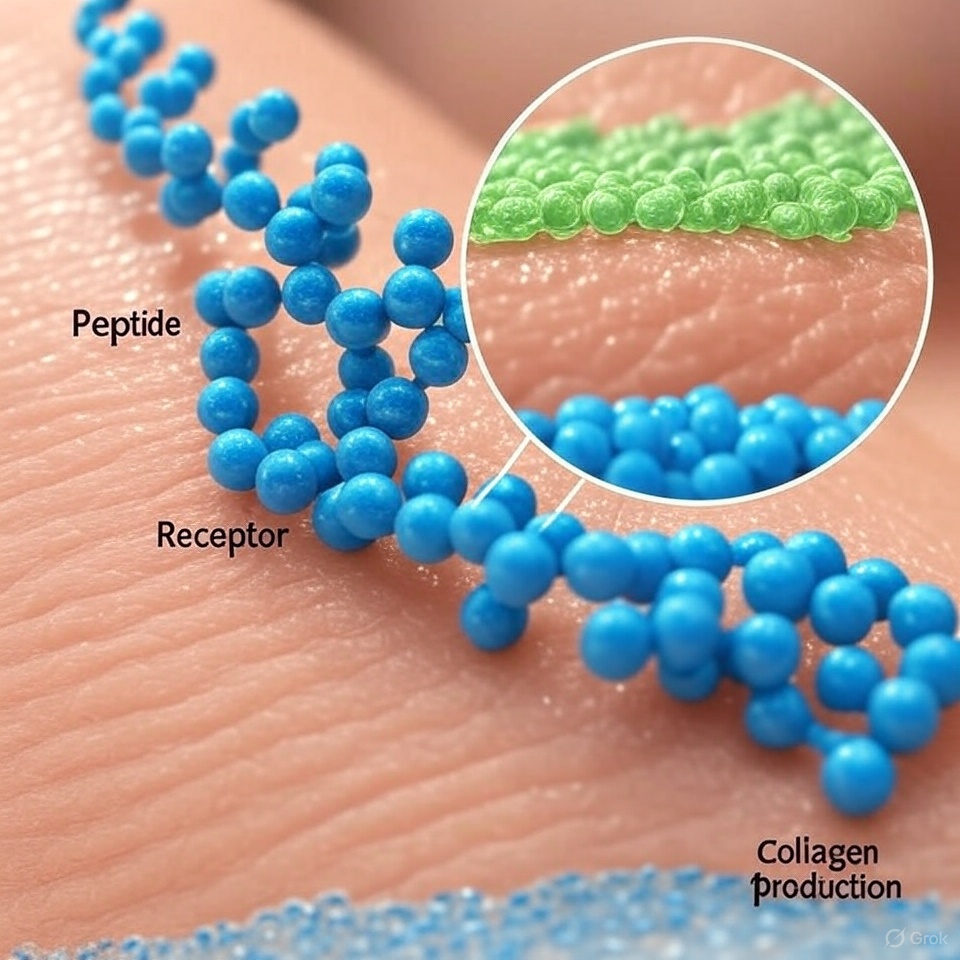
- Semaglutide remains a powerful research tool, not a lifestyle quick fix.
- If you’re studying or using peptides, approach with curiosity, caution, and a focus on data—not trends.
The Ozempic MDL shines a spotlight on the ripple effects of medical trends popularized by mass media. While lawsuits shake up the narrative, researchers must stay grounded: differentiate between research-grade compounds and marketed solutions, rely on evidence, and remember that legends don’t always emerge from lab-tested truth.